Breakthrough Creation of Novel Compound with Unique Mechanism of Action for Anti-Pancreatic Cancer Drug Development
Summary
Pancreatic cancer, notorious for its alarmingly low 5-year survival rate compared to other cancer types, demands urgent breakthroughs in treatment innovation. Led by Associate Professor Suresh Awale from the Institute of Natural Medicine, a collaborative research group comprising Professor Naoki Toyooka and Assistant Professor Takuya Okada from the Faculty of Engineering, Professor Tsutomu Fujii at the Faculty of Medicine from University of Toyama, and Lecturer Mitsuro Kanda from Nagoya University’s Graduate School of Medicine, has achieved a remarkable milestone. They have successfully created a groundbreaking compound that holds immense promise for pancreatic cancer treatment. Distinguished by its unique mechanism of action, distinct from existing anticancer medications, this compound opens up new avenues for chemotherapy in combating pancreatic cancer.
The momentous research findings were recently published online on May 31, 2023 (Wednesday) in the renowned Journal of Medicinal Chemistry, a leading journal in the field of medicinal chemistry.
Background of the Research
Pancreatic cancer, recognized as one of the most formidable solid tumors to combat, exhibits an exceptionally low 5-year survival rate compared to other types of cancer. Unlike other tumors, pancreatic tumors are typically characterized by limited blood vessel formation, resulting in restricted oxygen and nutrient availability within the tumor microenvironment. As a consequence, pancreatic cancer cells have evolved unique survival mechanisms by altering their energy metabolism, enabling them to thrive in an environment deprived of adequate oxygen and nutrients. However, conventional anticancer drugs such as gemcitabine and paclitaxel, designed to target rapidly proliferating cancer cells, prove ineffective in effectively countering these resilient pancreatic cancer cells. Moreover, the concerning issue of severe side effects associated with these drugs has intensified the demand for the development of novel treatment modalities based on alternative mechanisms of action capable of effectively targeting the microenvironment of pancreatic cancer.
In response to these challenges, Dr. Suresh Awale and his research group have been dedicated to developing compounds that selectively and potently exhibit toxicity against cancer cells in conditions that mimic the nutrient-deprived microenvironment of cancer. Their pioneering work aims to pave the way for new therapeutics specifically designed to combat pancreatic cancer.
Research Content and Achievements
The research group identified plumbagin, a component derived from the plant Plumbago auriculata, as a compound that selectively exhibits cytotoxicity against pancreatic cancer cells within a nutrient-deficient environment that mimics the conditions of the tumor microenvironment. Inspired by the chemical structure of plumbagin, the researchers successfully synthesized a groundbreaking compound, 3f, which exhibited enhanced potency and selectivity in terms of its cytotoxic effects compared to plumbagin. Through in-depth investigation of the compound’s mechanism of action, it was revealed that it induces cytotoxicity by inhibiting the Akt/mTOR signaling pathway. Encouragingly, animal experiments further demonstrated that compound 3f exhibited significant anticancer activity, effectively impeding tumor growth in a dose-dependent manner.
Future Developments
The research findings not only establish the immense promise of the novel plumbagin derivative, 3f, as a treatment for pancreatic cancer, utilizing a mechanism of action distinct from existing anticancer drugs, but also mark a significant breakthrough that expands the range of choices for pancreatic cancer chemotherapy. Moreover, the newly developed plumbagin derivative, 3f, holds the potential to contribute to overcoming the prevailing challenge of anticancer drug resistance by enabling combination therapies. Looking ahead, the research group will embark on clinical studies to validate the effectiveness of compound 3f as a viable treatment option for pancreatic cancer.
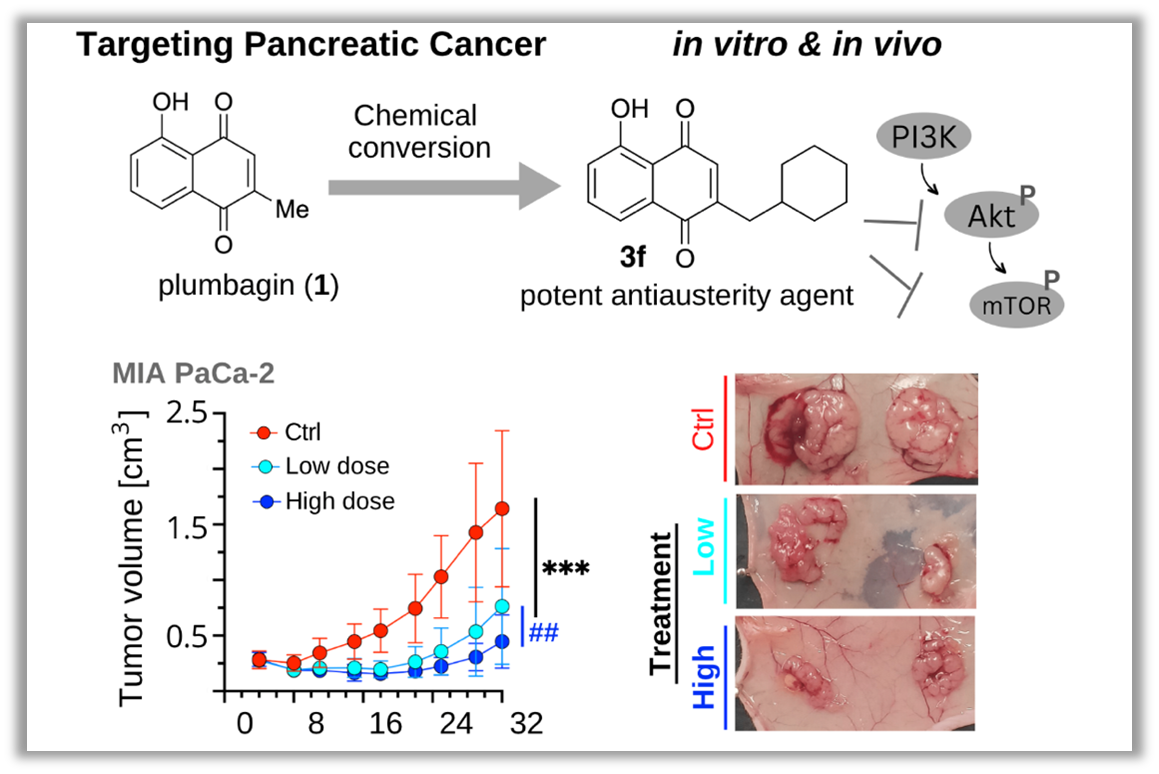 Figure 1. Research Outline
Figure 1. Research Outline
Publication Details
Title
Targeting Pancreatic Cancer with Novel Plumbagin Derivatives: Design, Synthesis, Molecular Mechanism, In Vitro and In Vivo Evaluation
Authors
Suresh Awale1,*, Hayato Baba2, Nguyen Duy Phan1,2, Min Jo Kim1, Juthamart Maneenet1, Koichi Sawaki3, Mitsuro Kanda3, Tomoyuki Okumura2, Tsutomu Fujii2, Takuya Okada4,5,*, Takahiro Maruyama5, Takahiro Okada5, Naoki Toyooka4,5,*
Affiliations
1 Natural Drug Discovery Laboratory, Institute of Natural Medicine, University of Toyama, 2630 Sugitani, Toyama 930-0194, Japan
2 Department of Surgery and Science, Graduate School of Medicine and Pharmaceutical Sciences, University of Toyama, Toyama 930-0194, Japan
3 Department of Gastroenterological Surgery, Graduate School of Medicine, Nagoya University, Nagoya 466-8560, Japan
4 Faculty of Engineering, University of Toyama, Toyama 930-8555, Japan
5 Graduate School of Science and Engineering, University of Toyama, Toyama 930-8555, Japan
Published in
Journal of Medicinal Chemistry
DOI
org/10.1021/acs.jmedchem.3c00394

