Success in Discovery of Key Compound for Development of Drug for Pompe Disease
Summary
Our research group led by Professor Atsushi Kato, Department of Hospital Pharmacy, University of Toyama, Professor Naoki Toyooka and Assistant Professor Takuya Okada, Faculty of Engineering, University of Toyama, and Professor Nobutada Tanaka, School of Pharmacy, Kitasato University, has successfully created an extremely promising compound that is a key to developing drugs for Pompe disease, a type of lysosomal disease, a nationally designated incurable disease. In this study, we have shown that chemical modification of the nitrogen atom of an iminosugar that exhibits a certain affinity for the enzyme responsible for Pompe disease can form a new strong interaction with the lipophilic pocket near the active site, and that the affinity was dramatically improved. Our results are expected to lead to the creation of more versatile and practical candidate compounds for Pompe disease, which has been considered difficult to treat due to its diverse mutations.
Our groundbreaking research entitled “Design and pharmacological chaperone effects of N-(4′-phenylbutyl)-DAB derivatives targeting the lipophilic pocket of lysosomal acid α-glucosidase” was published online June 14, 2023 in Journal of Medicinal Chemistry, the prestigious journal of the American Chemical Society in the field of medicinal chemistry.
Research Content and Achievements
Pompe disease, a type of lysosomal disease, is a disease in which glycogen abnormally accumulates in cells due to a deficiency of lysosomal acidic α-glucosidase, a sugar hydrolyzing enzyme (“glycosidase”). An effective small molecule drug for this disease is 1-deoxynojirimycin (DNJ), which is currently in clinical trials. DNJ has a structure very similar to glucose. Thus, “homology with sugar,” i.e., mimicking the structure of sugar, has been considered to be an effective means of compound design for small molecule drugs for this disease.
In this study, we focused on 1,4-dideoxy-1,4-imino-D-arabinitol (DAB), a naturally occurring sugar-like compound that exhibits inhibitory activity against a variety of glycosidases, but has selectivity problems and moderate inhibitory potency. Therefore, DAB was not expected to be used as a drug at all. We have shown that introduction of various substituent groups on the nitrogen atom of DAB, especially a phenylbutyl group, dramatically increases activity and improves selectivity, although homology to sugars is reduced. In particular, compound 5g bearing a (p-trifluoromethyl)phenylbutyl group on the nitrogen atom of DAB showed the potential to increase the activity of lysosomal acidic α-glucosidase in Pompe disease patients, an effect comparable to that of DNJ currently in clinical trials.
The development of drugs to treat nationally designated incurable diseases such as Pompe disease has lagged behind pharmaceutical manufacturers due to the small number of patients. Currently, enzyme replacement therapy, in which the defective enzyme is replaced externally, is a practical treatment for Pompe disease, but it requires intravenous infusion of an enzyme preparation once every two weeks, and because the treatment is lifelong, the cost is high, placing a heavy burden on patients. In addition, the continuous intravenous infusion of highly concentrated enzymes has the disadvantage of gradual loss of efficacy. Compound 5g found in this study is a small molecule and can be orally administered. It is expected to reduce the burden on patients and to have synergistic therapeutic effects when used in combination with enzyme preparations. The efficacy of 5g as a therapeutic agent for Pompe disease will be verified through clinical studies. Another important point of this study is the discovery of binding pockets of compounds in the enzyme that have been previously overlooked, through repeated docking simulations, structure-activity relationships, and experiments using patient-derived cells. Based on this discovery, it is highly likely that more potent candidate compounds will be found in the future.
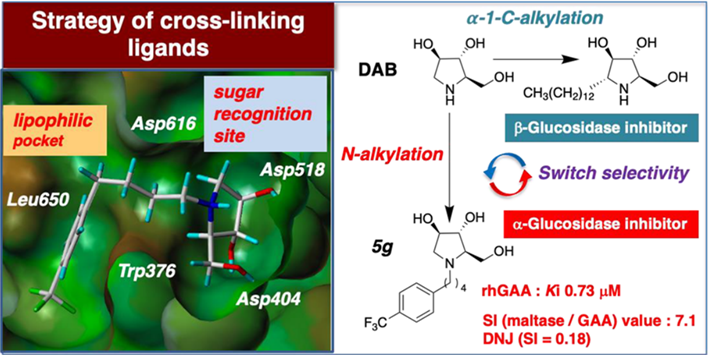 Figure Graphical abstract of our research
Figure Graphical abstract of our research
Publication Details
Title
Design and pharmacological chaperone effects of N-(4′-phenylbutyl)-DAB derivatives targeting the lipophilic pocket of lysosomal acid α-glucosidase
Authors
Atsushi Kato1,*, Izumi Nakagome2, Maki Kise1, Kousuke Yoshimura1, Nobutada Tanaka2,*, Robert J. Nash3, George W. J. Fleet4, Yota Kobayashi5, Hayato Ikeda6, Takuya Okada5,6,*, Naoki Toyooka5,6
Affiliations
1 Department of Hospital Pharmacy, University of Toyama, 2630 Sugitani, Toyama 930-0194, Japan.
2 School of Pharmacy, Kitasato University, Tokyo 108-8641, Japan.
3 Institute of Biological, Environmental and Rural Sciences / Phytoquest Limited, Plas Gogerddan, Aberystwyth, Ceredigion, SY23 3EB, United Kingdom.
4 Chemistry Research Laboratory, Department of Chemistry, University of Oxford, Oxford, OX1 3TA, United Kingdom.
5 Graduate School of Pharma-Medical Sciences, University of Toyama, Toyama 930-8555, Japan.
6 Faculty of Engineering, University of Toyama, Toyama 930-8555, Japan.
Published in
Journal of Medicinal Chemistry
DOI
org/10.1021/acs.jmedchem.3c00637

