Transcription Factor SOX10 regulates melanoma immunogenicity through IRF4-IRF1 axis
The research group of Associate Prof. Satoru Yokoyama (Dept. Cancer Cell Biology, Faculty of Pharmaceutical Sciences, University of Toyama), and Prof. Yoshihiro Hayakawa (Institute of Natural Medicine, University of Toyama) investigated that a transcription factor SOX10 regulates IRF1 independent on JAK activity. In addition, histone deacetylase inhibitors can enhance the efficacy of anti-PD-1 antibody, an immune checkpoint inhibitor, through SOX10 suppression in a clinically-relevant murine melanoma model. These findings will open up the therapeutic opportunity even in non-responders to immune checkpoint inhibitors.
Background
For immunotherapy, anti-Programmed Death 1 (PD-1) monoclonal antibody shows long-lasting clinical responses in some melanoma patients; however, a significant faction of patients are non-responders to anti-PD-1 antibody. Tumor immunogenicity, which is defined by intrinsic properties of cancer cell itself or by tumor microenvironment, is critically important for the efficacy of anti-PD-1 antibody. Especially, the signaling of IFN-g in cancer cells, which is secreted by immune cells, plays a significant role for the responsiveness to anti-PD-1 antibody. By lacking responsiveness to IFN-g with loss-of-function mutations of JAK1/2, the immunogenicity of cancer cells is significantly impaired due to non-induction of IRF1, a master regulator for tumor immunogenicity. Therefore, an understanding of other by-passing pathways to activate IFN-g signaling in cancer cells is clinically important for the success of cancer immunotherapy.
Results
Here, Yokoyama et al. demonstrated that the transcription factor SOX10 hinders immunogenicity of melanoma through the IRF4-IRF1 axis. Using both genetic and pharmacological approaches, the authors also demonstrated that SOX10 represses IRF1 transcription through the direct induction of a negative IRF1 regulator, IRF4. Importantly, the SOX10-IRF4-IRF1 axis regulated PD-L1 expression independently from the JAK-STAT signaling pathway, and the suppression of SOX10 increased the efficacy of combination therapy of anti-PD-1 antibody and HDAC inhibitor against a clinically-relevant melanoma model. Thus, the SOX10-IRF4-IRF1 axis is a potential target to immunologically warm up melanoma patients who are in “cold” non-responsive state in their tumor microenvironment by by-passing JAK-STAT signaling (Fig. 1).
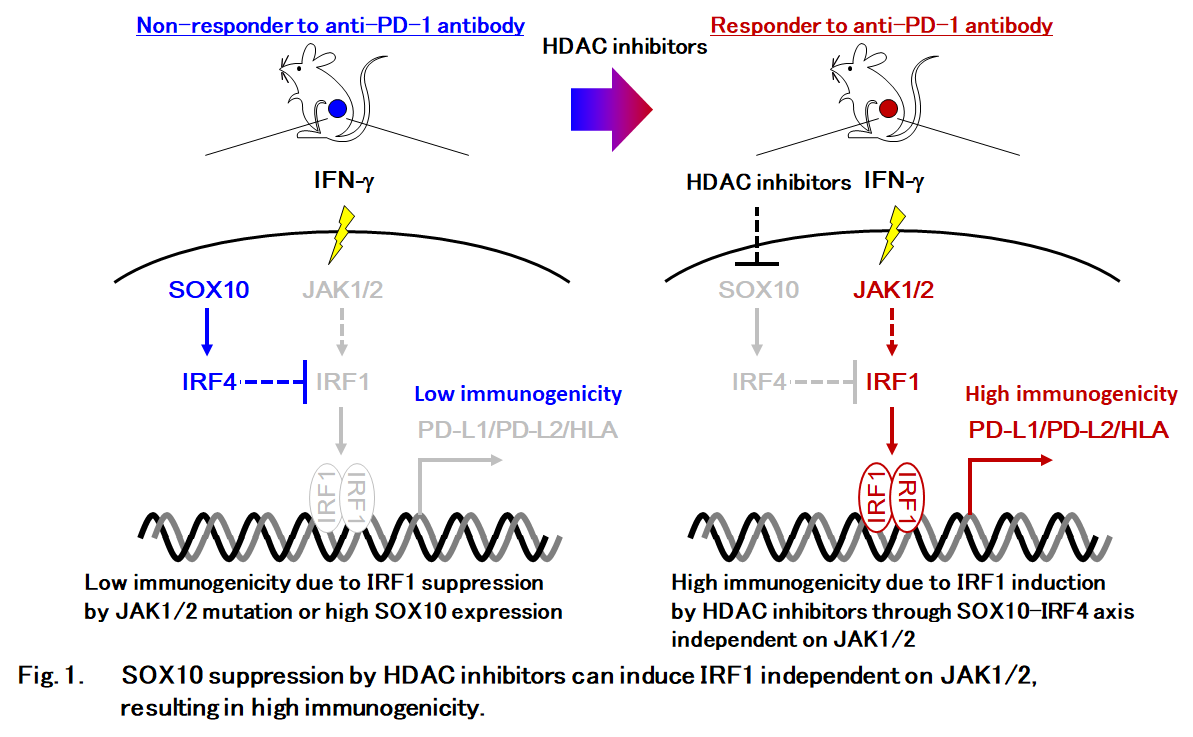
Significance of the research and future prospects
In this study, Yokoyama et al. identified that SOX10 suppression by histone deacetylase inhibitors can induce melanoma immunogenicity through IRF4-IRF1 axis but not through JAK1 or JAK2, and can enhance the efficacy of anit-PD-1 antibody. In other words, JAK-independent by-passing pathway to induce IRF1 can enhance the efficacy of anit-PD-1 antibody in melanoma patients. Given JAK1/2 mutation in other cancer subtypes than melanoma, identification of a JAK-independent pathway to induce IRF1 will provide hope for many cancer patients.
Publication information
Journal
Cancer Research
Publication Date
Nov. 2nd, 2021
Title
SOX10 regulates melanoma immunogenicity through an IRF4-IRF1 axis.
Authors
Yokoyama S, Takahashi A, Kikuchi R, Nishibu S, Lo JA, Hejna M, Moon WM, Kato S, Zhou Y, Hodi FS, Song JS, Sakurai H, Fisher DE, Hayakawa Y.
URL
SOX10 regulates melanoma immunogenicity through an IRF4-IRF1 axis | Cancer Research
DOI
10.1158/0008-5472.CAN-21-2078

