Skeletal muscle controls the onset of dementia ! ~Unbeneficial molecule is secreted from atrophied skeletal muscle and is delivered to the brain
Summary
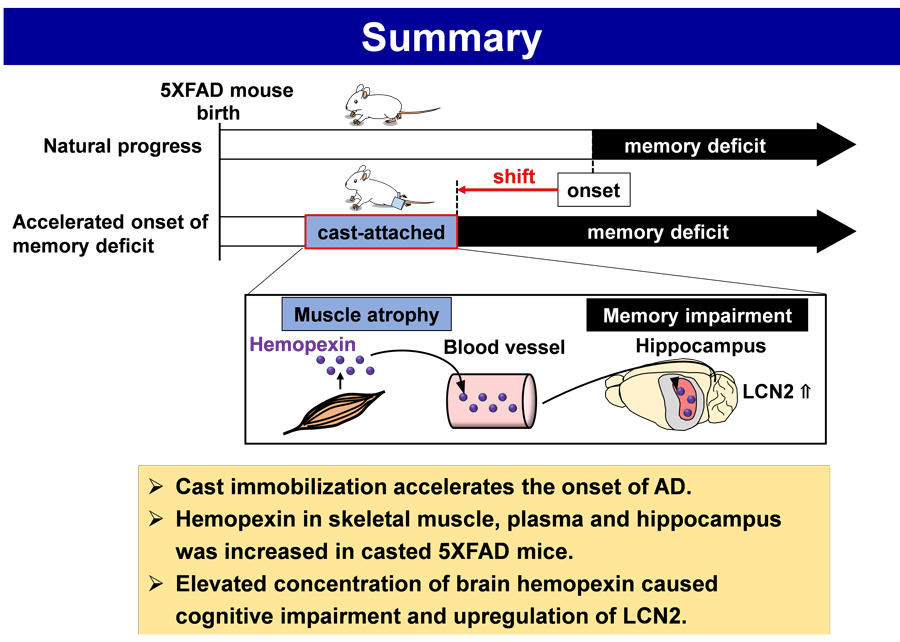
Pre-symptomatic young 5XFAD (Alzheimer’s disease (AD) model) or non-transgenic wildtype mice were used. Casting (immobilization of hindlimbs) for 2 weeks reduced skeletal muscle weight. Object recognition memory in the cast-attached 5XFAD mice was impaired than that in age-matched wildtype and non-cast 5XFAD mice. From conditioned medium of organ cultured atrophied muscles, we identified hemopexin as the most abundantly secreted protein. Hemopexin levels were increased by casting in the skeletal muscle, plasma, and hippocampus of cast-attached 5XFAD mice. Continuous intracerebroventricular infusion of hemopexin for 2 weeks induced memory deficits in young 5XFAD mice. Lipocalin-2 mRNA, neuroinflammation-associated factor, was increased in the hippocampus in hemopexin-infused 5XFAD mice.
Key findings
The skeletal muscle atrophy has an unbeneficial impact on the occurrence of memory impairment in young mice, which is mediated by the muscle-origin hemopexin and hemopexin-driven neuroinflammation.
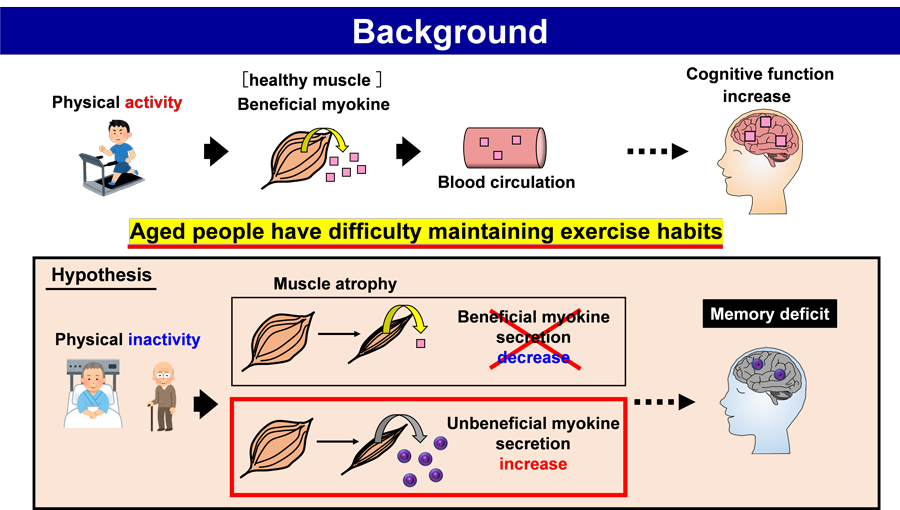
Background and overview
Several epidemiological studies suggest that traumatic brain injury, lifestyle-related disease, and smoking are major risk factors for AD. However, risk-reducing factors for AD, such as long education period and physical exercise, are also indicated. Age-associated muscle atrophy is known as sarcopenia. The relationship between sarcopenia and cognitive function has been shown to be negatively correlated by the cross-sectional study of healthy old men and meta-analysis. Regarding hospitalization-related disorders, a cross-sectional study of hospitalized aged adults investigating relationship between the prevalence of sarcopenia and cognitive impairment showed that the both of decrease in physical activity and cognitive decline were associated with hospitalization. Although those studies suggest the relationship among aging, skeletal muscle atrophy, and cognitive decline, no obvious evidences show that skeletal muscle atrophy directly induces cognitive impairment. As physical exercise is difficult to perform with increasing age, we hypothesized that a decline in physical movement may be a cause of age-associated lowering of the brain function. A decline in physical exercise leads to the loss of skeletal muscle. We hypothesized that sarcopenia induced the onset of AD, and therefore, we aimed to clarify that the skeletal muscle atrophy triggered a decline in brain function in AD.
We hypothesized that some molecules traveling from the atrophied skeletal muscles to the brain may affect the brain function. Such as “unbeneficial myokine for brain function” has not been identified yet. Therefore, this study aimed to elucidate the phenomenon of the skeletal muscle atrophy-induced acceleration of AD onset and its molecular mechanism.
This study revealed that the disuse-induced skeletal muscle atrophy shifted the onset of memory dysfunction earlier without increasing the deposition of Aβ in young mice model of AD. The atrophied muscles secreted hemopexin, and hemopexin was transported to the brain, possibly via systemic circulation. Moreover, increased levels of hemopexin in the brain or cast immobilization-induced muscle atrophy similarly impaired memory function and elevated the expression of neuroinflammatory factor, lipocalin-2 (LCN2) in the hippocampal CA3 neurons.
Article
Skeletal muscle atrophy-induced hemopexin accelerates onset of cognitive impairment in Alzheimer’s disease
Authors
Tsukasa Nagase, Chihiro Tohda.
Journal
Journal of Cachexia, Sarcopenia and Muscle
Publication date
17 October, 2021

