Metal 3D printing technology for functional integration of catalytic system
Summary
Mechanical properties and geometries of printed products have been extensively studied in metal 3D printing. However, chemical properties and catalytic functions, introduced by metal 3D printing itself, are rarely mentioned. Here we show that metal 3D printing products themselves can simultaneously serve as chemical reactors and catalysts (denoted as selfcatalytic reactor or SCR) for direct conversion of C1 molecules (including CO, CO2 and CH4) into high value-added chemicals. The Fe-SCR and Co-SCR successfully catalyze synthesis of liquid fuel from Fischer-Tropsch synthesis and CO2 hydrogenation; the Ni-SCR efficiently produces syngas (CO/H2) by CO2 reforming of CH4.
Research background
Catalysts and reactors are two essential elements of traditional catalytic systems. Until now, the researches of catalysts and reactors are still two different directions. Few studies have succeeded in functional integration of catalyst and reactor to effectively control chemical reaction. Therefore, there is a great need to develop their functional integration and synergies for future catalytic systems to realize superior chemical synthesis. On this basis, our integrated design, that is the printed self-catalytic reactors (SCRs) in combination with various catalytic functions (Fig. 1a, b), can further minimize the costs and reactor sizes dramatically, and improve the energy efficiency. Herein, we design and manufacture three kinds of SCRs (FeSCR, Co-SCR, and Ni-SCR) to realize direct conversion of C1 molecules (including CO, CO2, and CH4) into high value-added chemicals.
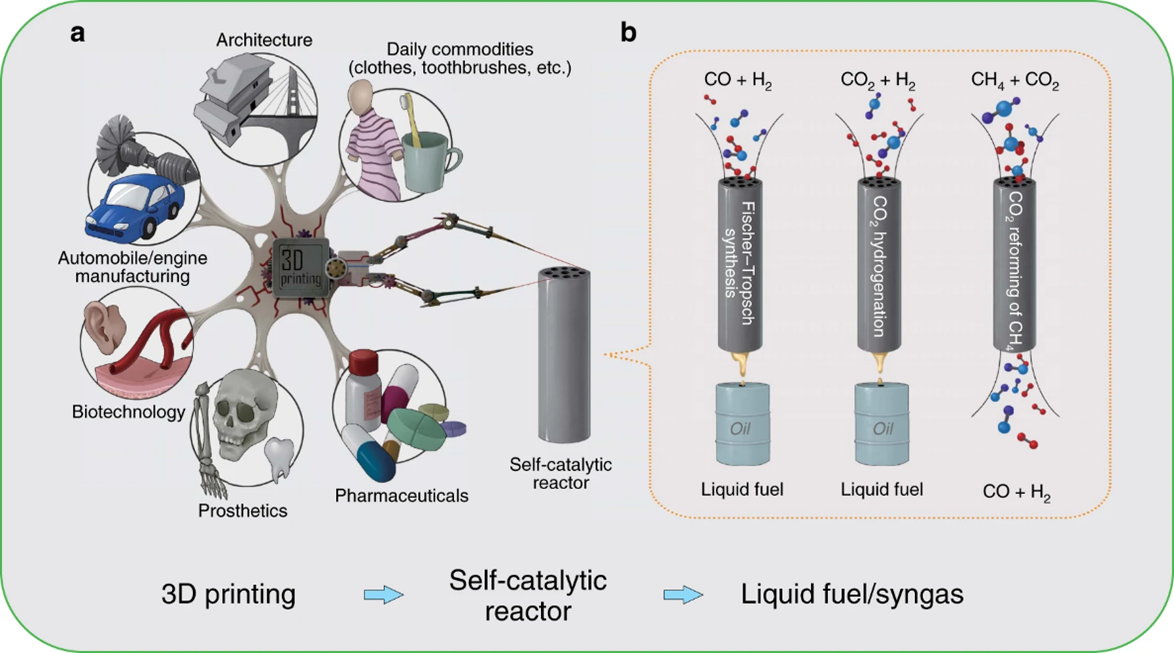
Fig. 1 3D printing for self-catalytic reactor (SCR) and other typical applications.
(a) 3D printing for SCR and other typical applications.
(b) The SCR for Fischer–Tropsch (FT) synthesis, CO2 hydrogenation, and CO2 reforming of CH4 (DRM).
Results
We have successfully designed three kinds of SCRs (Fe-SCR, Co-SCR, and Ni-SCR) via metal 3D printing technology. The C1 catalytic results demonstrated that they can effectively resist the harsh reaction conditions, such as high temperature and high pressure. The Fe-SCR and Co-SCR exhibited excellent performance for FT synthesis and CO2 hydrogenation to synthesize liquid fuel. The Ni-SCR displayed high conversions and ideal ratio of H2/CO for CO2 reforming of CH4. Further, the geometrical studies of the Co-SCRs clearly revealed that different printing structures can dramatically tune their catalytic functions.
Future direction
We expect that this self-catalytic design can stimulate new developments for 3D printing technologies, and wide applications in chemistry, energy, pharmacy, material synthesis, machinery manufacturing, etc.
Original article information
Journal
Nature Communications
Title
Metal 3D printing technology for functional integration of catalytic system
Authors
Qinhong Wei1,2,7, Hangjie Li1,7, Guoguo Liu1,7, Yingluo He1, Yang Wang1, Yen Ee Tan1, Ding Wang3, Xiaobo Peng4,5, Guohui Yang1,6 & Noritatsu Tsubaki1
1Department of Applied Chemistry, School of Engineering, University of Toyama, Gofuku 3190, Toyama 930-8555, Japan.
2Department of Chemical Engineering, School of Petrochemical Technology and Energy Engineering, Zhejiang Ocean University, Zhoushan 316022, China.
3School of Material Science & Engineering, University of Shanghai for Science and Technology, Shanghai 200093, China.
4National Institute for Materials Science, 1-1 Namiki, Tsukuba, Ibaraki 305-0044, Japan.
5Key Laboratory of the Ministry of Education for Advanced Catalysis Materials, Institute of Physical Chemistry, Zhejiang Normal University, Jinhua 321004, China.
6State Key Laboratory of Coal Conversion, Institute of Coal Chemistry, Chinese Academy of Sciences, Taiyuan 030001, China.
7These authors contributed equally: Qinhong Wei, Hangjie Li, Guoguo Liu.

