Ammonia pools in zeolites for direct fabrication of catalytic centers
Summary
Reduction process is a key step to fabricate metal-zeolite catalysts in catalytic synthesis. However, because of the strong interaction force, metal oxides in zeolites are very difficult to be reduced. Existing reduction technologies are always energy-intensive, and inevitably cause the agglomeration of metallic particles in metal-zeolite catalysts or destroy zeolite structure in severe cases. Herein, we disclose that zeolites after ion exchange of ammonium have an interesting and unexpected self-reducing feature. It can accurately control the reduction of metal-zeolite catalysts, via in situ ammonia production from ‘ammonia pools’, meanwhile, restrains the growth of the size of metals.
Research background
Up to now, several kinds of zeolites have been applied as catalysts in the oil refining and petrochemical processes benefitting from their superior thermostability, renewability, and catalytic selectivity1. Coordinating with metals, the role of zeolites in industrial catalysis is being further enhanced. Zeolite-supported transition metal catalysts, as a class of bifunctional heterogeneous catalysts, have been successfully applied in industrial catalytic processes, such as methanol to aromatics, methanol to olefins, ammonia selective catalytic reduction, and so on. Nevertheless, the fabrication process of metal-zeolite catalysts always involves reducing metal precursors under the reducing gas (mainly including H2, CO, or syngas (CO + H2)) flow, which is very energy-intensive.
Here, we employ a series of zeolites as supports whose pore channels brim with ammonium ions (named ‘ammonia pools’), and disclose that these zeolites enable in situ reduction of supported metal species without using any additional reduction process (Fig. 1). We find that the ammonia molecules can be continuously released from ammonia pools during the calcination process of NH4-zeolites. Then, these ammonia molecules mildly and efficiently reduce supported metal oxides, while maintaining small sizes and high degree of dispersion for the metallic particles.
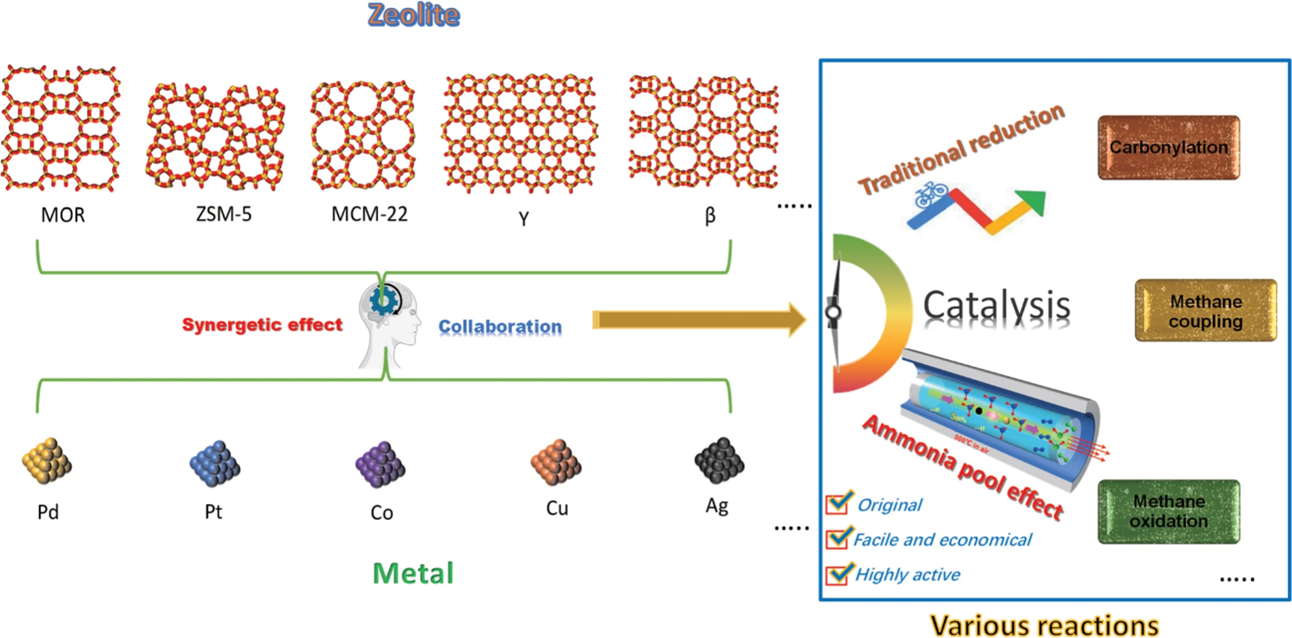
Fig. 1 The ammonia pool effect in different types of zeolites and metals for various catalytic reactions. Metal-zeolite catalysts, consisting of many base/ noble metals (Fe, Co, Ni, Cu, Pt, Ag, Au, and Pb) being incorporated in varied zeolites (as MOR, ZSM-5, MCM-22, Y, β), are rationally fabricated by ammonia pool effect to realize considerably better synergetic effect in a series of reactions involving carbonylation, methane coupling, and methane oxidation. The presented ammonia pool effect is original, facile, economical, and highly efficient.
Results
We presented a zeolite-specific self-reducing methodology, i.e., reduction via ammonia pool effect (abbreviated as APE), and proved this widely applied and convenient technology for efficient catalysis. The features of APE technology were disclosed based on a series of rationally designed characterizations and experiments. We found that the APE technology was not influenced by different structures and types of zeolites, and acted well on various metal ions. Moreover, compared with traditional reduction manners, the superior APE technology fabricated smaller and more dispersed active metal particles in metal-zeolite catalysts. Most importantly, these catalysts further realized higher catalytic activity and selectivity than those obtained via traditional H2 reduction. We finally put forward an atmosphere-confined self-regulation mechanism and clearly explain this interesting but important phenomenon. This work uncovers a hidden specialty of zeolites.
Future direction
We anticipate that the APE strategy demonstrated here will be pivotal for energy-saving fabrication of highly active and stable catalysts, for conversion of important molecules (such as methane, syngas, dimethyl ether, etc.) to high value-added chemicals.
Reference
- 1. Primo, A. & Garcia, H. Zeolites as catalysts in oil refining. Chem. Soc. Rev. 43, 7548–7561 (2014).
Original article information
Journal
Nature Communications
Title
Ammonia pools in zeolites for direct fabrication of catalytic centers
Authors
Jie Yao1,6, Yingluo He1,6, Yan Zeng1,6, Xiaobo Feng1,2, Jiaqi Fan1, Shoya Komiyama1, Xiaojing Yong3, Wei Zhang3, Tiejian Zhao3, Zhongshan Guo3, Xiaobo Peng1,4, Guohui Yang1,5, Noritatsu Tsubaki1
1Department of Applied Chemistry, School of Engineering, University of Toyama, Gofuku 3190, Toyama 930-8555, Japan.
2 Jiangsu Province Engineering Research Center of Fine Utilization of Carbon Resources, China University of Mining & Technology, Xuzhou 221116 Jiangsu, China.
3National Energy Group Ningxia Coal Industry Co., Ltd., No. 168 Beijing Middle Road, Yinchuan, China.
4National Engineering Research Center of Chemical Fertilizer Catalyst, Fuzhou University, Fuzhou 350002 Fujian, China.
5State Key Laboratory of Coal Conversion, Institute of Coal Chemistry, Chinese Academy of Sciences, Shanxi 030001 Taiyuan, China.
6These authors contributed equally: Jie Yao, Yingluo He, Yan Zeng.

