Success in Creation of Novel Compounds with Potential for Use in the Treatment of Intractable Amyloid Disease
Summary
The research group led by Professor Naoki Toyooka and Assistant Professor Takuya Okada of Faculty of Engineering, University of Toyama, and Professor Mineyuki Mizuguchi and Assistant Professor Takeshi Yokoyama of Faculty of Pharmaceutical Sciences of the same university have successfully created a new compound that could be applied to a new therapeutic agent for amyloid disease, a nationally designated intractable disease. The newly discovered compound is an analog of benzbromarone, a drug that promotes uric acid excretion. Transthyretin (TTR) is a stable protein, but under certain conditions or due to genetic factors, it forms amyloid fibrils, which are deposited in peripheral nerves, heart, and eyes, causing serious functional disorders. This is called amyloid disease and is designated as an intractable disease, but the only effective small molecule drug for this disease is tafamidis (Vindakel). The newly synthesized benzbromarone derivatives bind more strongly than tafamidis and strongly inhibit amyloid fibril formation and are expected to be extremely promising as a new therapeutic agent for amyloid disease.
The research results were published online in the Journal of Medicinal Chemistry, an authoritative American Chemical Society journal in the field of medicinal chemistry, on Friday, April 26, 2024.
Research Background and Results
Transthyretin (TTR) is a tetrameric protein synthesized mainly in the liver and is responsible for the transport of thyroxine and retinol. Normally, TTR is a stable protein, but when the tetramer dissociates and aggregates into a monomer under certain conditions or due to genetic factors, it forms insoluble amyloid fibrils that are deposited in peripheral nerves, heart, and eyes, causing serious functional disorders. This is called amyloid disease and is designated as an intractable disease (Figure 1).
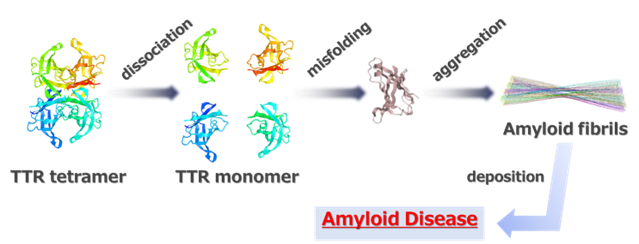
Figure 1 Pathogenesis of Amyloid Disease
Since approximately 95% of TTR is synthesized in the liver, liver transplantation is an effective treatment for this disease, but the shortage of donors is a major problem. Currently, tafamidis (Vindakel) is in clinical use as an effective small molecule drug, but it may not sufficiently inhibit disease progression. Therefore, the development of new therapeutic agents that are more effective than tafamidis is highly desirable.
In 2023, our research group demonstrated that benzbromarone and benziodarone, both uric acid excretion stimulants, bind specifically to TTR in plasma and exhibit favorable amyloid fibril inhibitory effects. In this study, we designed new compounds based on benzbromarone and studied their synthesis. As a result of synthesis of about 50 new benzbromarone derivatives, we succeeded in creating compounds with strong amyloid fibril inhibitory effects. By comparing the chemical structures among the synthesized derivatives, we succeeded in identifying the substructures that are essential for the activity (Figure 2).
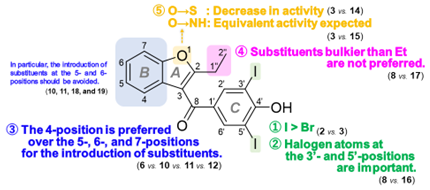
Figure 2 Structure-activity relationship of the benzbromarone derivatives
Human plasma contains not only TTR but also various other proteins. As a result of an investigation into the specific binding of the synthesized compound to TTR, it was demonstrated that the synthesized compound shows selective and strong binding to TTR in human plasma. Therefore, the novel benzbromarone derivative we synthesized is expected to be extremely promising as a new therapeutic agent for amyloid disease.
The novel benzbromarone derivatives discovered in this study have the potential to become new amyloid disease drugs that are more effective than tafamidis. In the future, we intend to verify the efficacy of the developed compounds as a treatment for amyloid disease through clinical trials.
Original article information
Title
Development of benziodarone analogues with enhanced potency for selective binding to transthyretin in human plasma
Authors
Mineyuki Mizuguchi*1, Yusuke Nakagawa*2, Takeshi Yokoyama*1, Takuya Okada*2,*3, Kanako Fujii*4, Kanoko Takahashi*5, Nguyen Ngoc Thanh Luan*2, Yuko Nabeshima*1, Kayoko Kanamitsu*6, Shinsaku Nakagawa*7, Shiori Yamakawa*8, Mitsuharu Ueda*8, Yukio Ando*9, Naoki Toyooka*2,*3
*1.Faculty of Pharmaceutical Sciences, University of Toyama, Toyama, 930-0194, Japan.
*2.Graduate School of Innovative Life Science, University of Toyama, Toyama 930-8555, Japan.
*3.Faculty of Engineering, University of Toyama, Toyama 930-8555, Japan.
*4.Graduate School of Medicine and Pharmaceutical Sciences, University of Toyama, Toyama 930-0194, Japan.
*5.Graduate School of Pharma-Medical Sciences, University of Toyama, Toyama 930-8555, Japan.
*6.Graduate School of Pharmaceutical Sciences, the University of Tokyo, Tokyo 113-0033, Japan.
*7.Graduate School of Pharmaceutical Sciences, Osaka University, Osaka 565-0871, Japan.
*8.Department of Neurology, Graduate School of Medical Sciences, Kumamoto University, Kumamoto 860-8556, Japan.
*9.Faculty of Pharmaceutical Sciences, Nagasaki International University, Sasebo 859-3298, Japan.

